As the country prepares to enter a second lockdown from Thursday, there is a glimmer of hope after reports of vaccine DES will be announced. It is believed practices and Primary Care Network’s (PCN’s 0will be able to start administering the life-saving drug from the beginning of December. PCN’s are groups of practices working together to focus local patient care.
According to reports, practices and PCN’s will be asked to give the vaccine to over-85s and front line workers as early as next month.
Two vaccines are currently being prepared with on requiring two shots and needing to be kept a minus 70 degrees.
Teams will also deliver the potential life-saving vaccine to care homes and at-scale delivery centres.
An announcement on DES is imminent and could be announced next week, sources told Pulse.

COVID vaccine to be rolled out in December (Image: Getty)

The new vaccine to be rolled out in December (Image: Getty)
Last month, the NHS said it was looking to start the rollout of the vaccine from December, with sources putting the chances at ’50/50’.
But now, several sources have confirmed GPs and the whole of the NHS will be put on standby to start vaccinating the more vulnerable groups from next month.
There are currently two frontrunners in the development of a vaccine including Pfizer and Oxford/AstraZeneca.
Pfizer’s vaccine requires it to be kept at minus 70 degrees, while the other was shown to be more effective when administered in two doses.
READ MORE: Coronavirus vaccine developed, potentially 10 TIMES higher immunity

Sir Patrick Vallance (Image: Getty)
While it is a breakthrough in vaccine research, Sir Patrick Vallance has played down the chances of an effective drug to combat the deadly virus.
Over the weekend, AstraZeneca revealed the country’s health regulator has begun a review of the potential vaccine.
A spokesperson for the pharmaceutical company said: “We confirm the MHRA’s (Medicines and Healthcare Products Regulatory Agency) rolling review of our potential COVID-19 vaccine.”
During these reviews, regulators are able to see clinical data in real time and speak to drug makers on manufacturing processes and trials to accelerate the approval process.
DON’T MISS
Boris Johnson URGED to make coronavirus U-turn by Ex-Health Secretary [REVEAL]
Coronavirus: Study identifies genetic quirk that exaggerates reaction [INSIGHT]
Blair urges vaccine giant to ‘start injecting’ people with COVID cure [COMMENT]
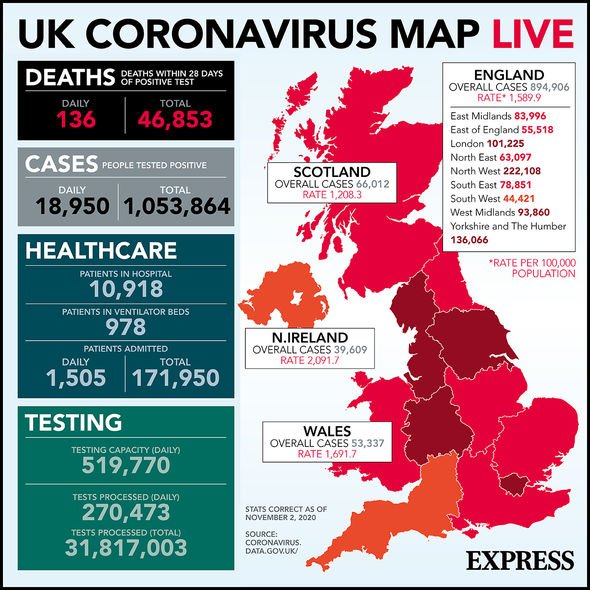
UK coronavirus cases mapped (Image: Express)
It is designed to help speed up evaluations of promising drugs and vaccines during a public health emergency.
On Friday, the MHRA were also reported to have begun another accelerated review for a vaccine from Pfizer Inc.
At the end of October, there was a breakthrough with the Astra-Zeneca/Oxford vaccine after analysis by Bristol University found greater evidence the vaccine triggers an immune response.
This study proved the vaccine is correctly programmed to replicate the “spike protein” associated with the deadly virus.

Boris Johnson announced lockdown for four weeks (Image: Getty)
The protein is what the body’s immune system will learn to attack if infected with coronavirus.
This news of a potential vaccine to be rolled out in December comes just days after Prime Minister Boris Johnson announced a second national lockdown to last four weeks.
Under the new lockdown measures, people are being told to stay at home unless they have a specific reason to leave such as work which cannot be done from home.
Meeting indoors or in private gardens will now be banned but individuals can meet one other person from another household outside in a public place.

GPs and PCN’s to give vaccine to over-85s (Image: Getty)
Pubs, bars, restaurants and non-essential retail across the country will close but takeaways will remain open.
Entertainment venues including gyms will also close again.
People are still allowed to form support bubbles.
Over the weekend, new infections of the deadly virus reached more than 20,000 bringing the overall total to more than a million confirmed cases.
The UK is now the ninth country to reach the milestone of more than a million confirmed cases.







