In a blow to the UK’s hopes of a vaccine, Ms Bingham also claimed the first batch of vaccines may also be “imperfect”. Even if the vaccines are proved to be successful, she claimed the immunity may not be permanent. Writing in the Lancet journal, she also stated the task force had been mainly focusing on drugs which trigger an immune response to those over 65.
She wrote: “We do not know that we will ever have a vaccine at all. It is important to guard against complacency and over-optimism.
“The first generation of vaccines is likely to be imperfect, and we should be prepared that they might not prevent infection but rather reduce symptoms, and, even then, might not work for everyone or for long.”
In a look to the future, she also warned the virus will be evolved and other pathogens may “pose future risks”.
Ms Bingham also claimed nations must work together to create a successful vaccine in order to protect the world.

Coronavirus vaccine may be imperfect (Image: GETTY)
Coronavirus: A vaccine could be rolled out before the end of the year (Image: GETTY)
She added: “There will not be one successful vaccine, or one single country, that is able to supply the world.
“We urgently need international cooperation to pool risks and costs, address barriers to access, and scale up the manufacturing capacity to produce sufficient doses to protect everyone at risk of SARS-CoV-2 infection globally.
“The SARS-CoV-2 virus is likely to evolve, and other zoonotic pathogens are likely to pose future risks. China, Europe, the USA, and the UK need to work together.”
Currently, the UK has secured access to six potential vaccines produced by AstraZeneca and the University of Oxford, BioNTech and Pfizer, Janssen, Novavax, GSK and Sanofi.
JUST IN: England second lockdown: Cabinet minister silent amid SAGE warning
Coronavirus: The drug from Oxford and AstraZeneca is leading the way (Image: GETTY)
As it stands, the vaccine being trialled by Oxford University and AstraZeneca is leading the group of possible drugs.
The developers of the drug, this week stated the potential vaccine, AZD1222, produced an immune response in all ages, not just the elderly.
The drug is currently undergoing a phase 3 clinical trials whereby they are tested on volunteers.
If it passes this stage, it will then be given approval to be manufactured on a wider scale.
DON’T MISS
When your tiredness could be a sign of COVID infection [Latest]
Health chiefs call for urgent action as Covid death toll hits 367 [Update]
EU coronavirus CHAOS as rioters across Europe protest new restrictions [Insight]
Coronavirus: The immunity may not be permanent (Image: PA)
Coronavirus: Kate Bingham receives vaccine (Image: GETTY/PA)
Indeed, if the vaccine proceeds through the trials, AstraZeneca believes some doses will be available before the turn of the year.
These doses, however, will not be on a widespread scale with the UK’s chief scientific advisor, Sir Patrick Vallance claiming a vaccine will not available until spring 2021.
A spokesperson for the country said: “It is encouraging to see immunogenicity responses were similar between older and younger adults and that reactogenicity was lower in older adults, where the Covid-19 disease severity is higher.
“The results further build the body of evidence for the safety and immunogenicity of AZD1222.
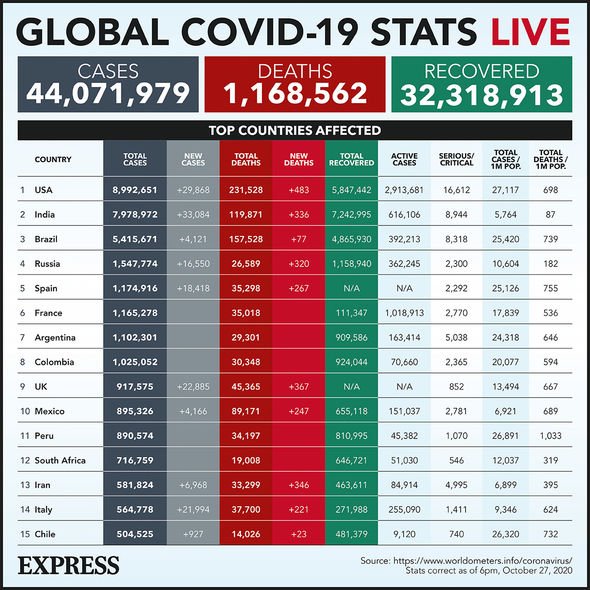
Coronavirus: Cases as of October 27 (Image: Express)
“We anticipate efficacy read-outs from phase 2/3 trials between now and the end of the year, and if approved within countries, doses of the potential vaccine could be available for use before the end of the year.”
The drug produced by Novavax has also proceeded to phase 3 trials at various sites across the UK.
The remaining vaccines of the group of six, will not be available until late 2021.

Coronavirus: Mr Hancock tempered expectations (Image: PA/GETTY)
Although not certain of its response, a vaccine is seen as the key the factor in the fight against coronavirus.
Health Secretary, Matt Hancock said on the chances of a vaccine: “It’s obviously something that we want to happen as soon as safely can be done.
“And as fast as safely can be done, but we are not there yet.”







